Exogenous hydrogen sulfide modulates metabolic responses of sugar and phenolic acid in naked oat leaves under saline-alkali stress
-
摘要: 为明确硫化氢信号对盐碱胁迫下植物代谢组的调控作用, 揭示其增强植物耐盐碱性的机理, 以裸燕麦(Avena nuda L.)为材料进行盆栽土培试验, 设置不添加盐碱和添加3.00 g·kg−1盐碱(摩尔比NaCl∶Na2SO4∶Na2CO3∶NaHCO3=12∶8∶1∶9)与裸燕麦抽穗期叶面喷施蒸馏水和50 µmol·L−1硫化氢供体硫氢化钠溶液交叉共4组处理, 利用超高效液相色谱-串联质谱技术结合正交偏最小二乘判别分析方法, 研究外源硫化氢对盐碱胁迫下裸燕麦叶片糖分解代谢产物水平、氧化还原平衡、酚酸含量和产量性状的影响。结果表明: 1)非盐碱条件下, 喷施硫氢化钠对裸燕麦叶片还原型谷胱甘肽/氧化型谷胱甘肽、还原型辅酶Ⅱ/氧化型辅酶Ⅱ、腺苷三磷酸含量和产量性状的影响微弱, 但显著上调柠檬酸、琥珀酸和6-磷酸葡萄糖酯含量, 显著下调葡萄糖-6-磷酸、丙酮酸、乳酸、α-酮戊二酸、谷氨酸、天冬酰胺、赤藓糖-4-磷酸、景天庚酮糖-7-磷酸含量; 盐碱胁迫导致裸燕麦叶片葡萄糖、葡萄糖-6-磷酸、果糖-6-磷酸、果糖-1,6-二磷酸、3-磷酸甘油醛、3-磷酸甘油酸、丙酮酸、乳酸、α-酮戊二酸、谷氨酸、谷氨酰胺、天冬酰胺、赤藓糖-4-磷酸、景天庚酮糖-7-磷酸、核糖-5-磷酸等糖酵解、三羧酸循环和戊糖磷酸途径中间代谢物及还原型谷胱甘肽、氧化型谷胱甘肽、还原型辅酶Ⅱ、氧化型辅酶Ⅱ含量显著降低, 而还原型谷胱甘肽/氧化型谷胱甘肽显著提高; 喷施硫氢化钠显著提高盐碱胁迫下裸燕麦叶片葡萄糖、果糖-6-磷酸、3-磷酸甘油酸、乳酸、α-酮戊二酸、延胡索酸、苹果酸、谷氨酰胺、6-磷酸葡萄糖酯、景天庚酮糖-7-磷酸含量, 显著降低天冬酰胺含量。2)喷施硫氢化钠显著下调非盐碱条件下裸燕麦叶片反式肉桂酸和丁香醛含量; 盐碱胁迫导致裸燕麦叶片反式肉桂酸含量显著降低, 而苯甲酸、对香豆酸和反式阿魏酸含量显著提高; 喷施硫氢化钠显著提高盐碱胁迫下裸燕麦叶片4-羟基苯甲酸和香草醛含量, 显著降低水杨酸、芥子酸含量。3)喷施硫氢化钠对盐碱胁迫造成的裸燕麦穗粒数量和籽粒产量下降具有显著缓解作用, 但对穗数量、穗铃数量、千粒重量和生物学产量的影响不显著。由此表明, 外源硫化氢参与裸燕麦糖分解代谢和酚酸水平调控, 能够增强裸燕麦耐受盐碱胁迫的能力, 它对糖分解途径有机酸水平的提升作用和酚酸独特的调节效应可能在其增强裸燕麦耐盐碱性中发挥着重要作用。Abstract: In order to clarify the regulatory effect of hydrogen sulfide signaling on the plant metabolome under saline-alkali stress and to reveal its mechanism of enhancing plant saline-alkali tolerance, a pot experiment was conducted with naked oat (Avena nude) as the material. Four treatments were applied to potted naked oat plants in a 2 × 2 factorial combination, including 0 or 3.00 g·kg−1 saline-alkali (molar ratio of NaCl∶Na2SO4∶Na2CO3∶NaHCO3 at 12∶8∶1∶9) added to the potting soil and spraying with distilled water or 50 µmol·L−1 sodium hydrosulfide (a hydrogen sulfide donor) on leaves at the heading stage. The effects of exogenous hydrogen sulfide on glycolytic metabolite levels, redox balance, and phenolic acid content in leaves and on the yield traits of naked oats under the four treatments were investigated using ultra-performance liquid chromatography-tandem mass spectrometry technology combined with orthogonal partial least squares discriminant analysis. Under non-saline-alkali conditions, sodium hydrosulfide application did not have a significant effect on the ratios of (reduced glutathione)/(oxidized glutathione) and (reduced coenzyme Ⅱ)/(oxidized coenzyme Ⅱ), adenosine triphosphate content in leaves, and yield traits of naked oats; however, the levels of citrate, succinate, and 6-phosphogluconolactone were significantly upregulated and those of glucose-6-phosphate, pyruvate, lactate, α-ketoglutaric acid, glutamate, asparagine, erythrose-4-phosphate, and sedoheptulose-7-phosphate were significantly downregulated in the leaves. Saline-alkali stress significantly reduced the levels of glucose, glucose-6-phosphate, fructose-6-phosphate, fructose-1,6-diphosphate, 3-phosphate glyceraldehydes, 3-phosphoglyceric acid, pyruvate, lactate, α-ketoglutaric acid, glutamate, glutamine, asparagine, erythrose-4-phosphate, sedoheptulose-7-phosphate, ribose-5-phosphate, reduced glutathione, oxidized glutathione, reduced coenzyme Ⅱ, and oxidized coenzyme Ⅱ in the leaves of naked oats; whereas the ratio of reduced glutathione to oxidized glutathione was increased significantly. Spraying with sodium hydrosulfide significantly increased the levels of glucose, fructose-6-phosphate, 3-phosphoglyceric acid, lactate, α-ketoglutaric acid, fumarate, malate, glutamine, 6-phosphogluconolactone, and sedoheptulose-7-phosphate in the leaves of naked oats under saline-alkali stress, and significantly decreased the asparagine content. The levels of trans-cinnamic acid and syringaldehyde in the leaves of naked oats under non-saline-alkali conditions were significantly decreased by spraying with sodium hydrosulfide. Saline-alkali stress significantly reduced the content of trans-cinnamic acid in the leaves of naked oats, and markedly increased the levels of benzoic acid, p-hydroxycinnamic acid, and trans-ferulic acid. Spraying with sodium hydrosulfide significantly increased the levels of 4-hydroxybenzoic acid and vanillin in the leaves of naked oats under saline-alkali stress, and remarkably decreased the levels of salicylic acid and 4-hydroxy-3,5-dimethoxycinnamic acid.There was no significant increase in spike number, spike boll number, thousand-grain weight, and biological yield of naked oats under saline-alkali stress as a result of spraying with sodium hydrosulfide; however, sodium hydrosulfide significantly alleviated the decrease in spike grain number and grain yield induced by saline-alkali stress. These results indicate that exogenous hydrogen sulfide participates in the regulation of sugar catabolism and phenolic acid levels in naked oats, which can enhance the saline-alkali tolerance of naked oats. The increasing effect of exogenous hydrogen sulfide on organic acid levels in the sugar decomposition pathway and the unique regulatory effect on phenolic acids may play an important role in enhancing the saline-alkali tolerance of naked oats.
-
-
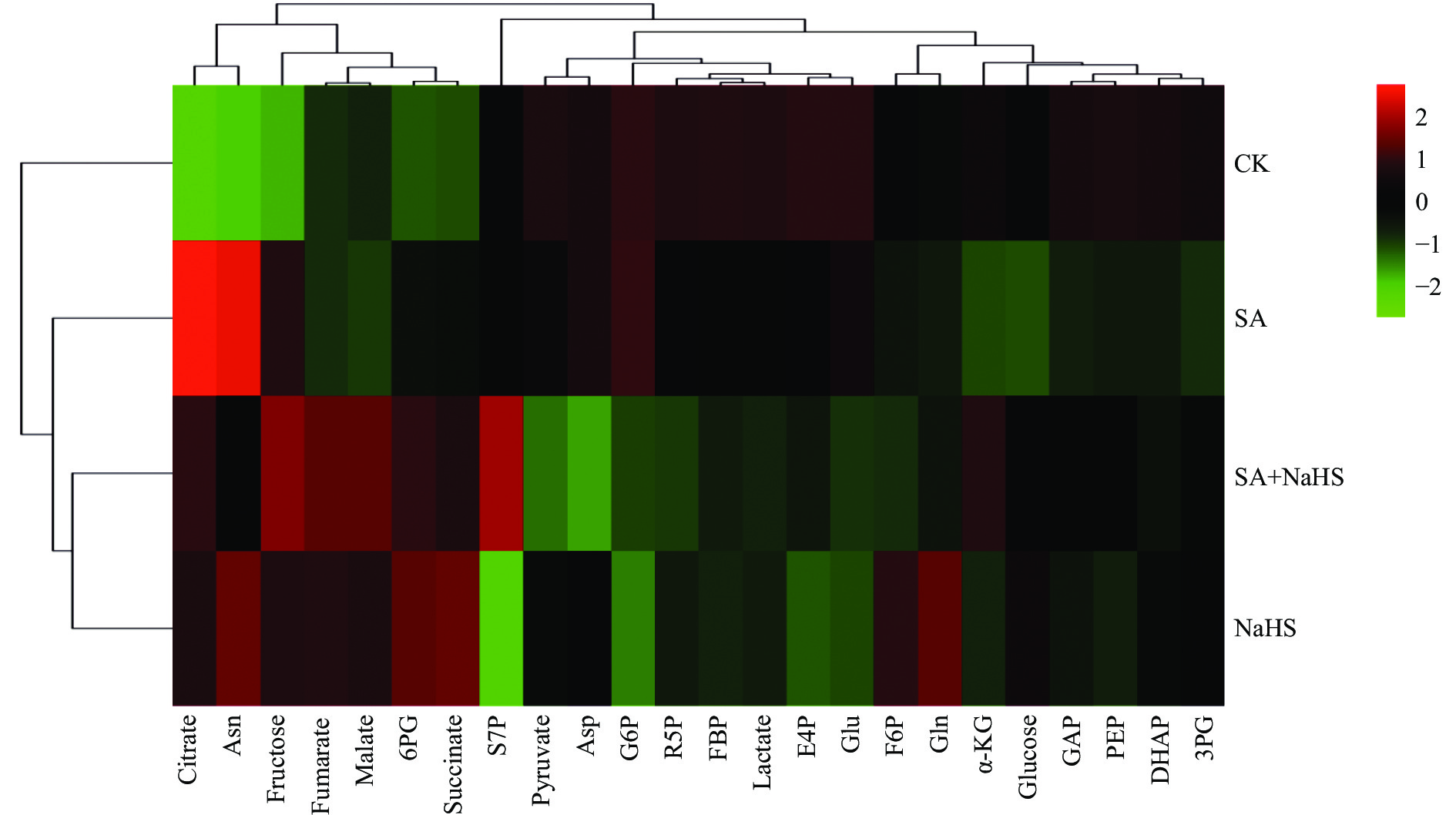
图 1 不同外源硫化氢喷施和盐碱胁迫处理下裸燕麦叶片糖分解代谢物聚类热图分析
CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。Citrate: 柠檬酸; Asn: 天冬酰胺; Fructose: 果糖; Fumarate: 延胡索酸; Malate: 苹果酸; 6PG: 6-磷酸葡萄糖酯; Succinate: 琥珀酸; S7P: 景天庚酮糖-7-磷酸; Pyruvate: 丙酮酸; Asp: 天冬氨酸; G6P: 葡萄糖-6-磷酸; R5P: 核糖-5-磷酸; FBP: 果糖-1,6-二磷酸; Lactate: 乳酸; E4P: 赤藓糖-4-磷酸; Glu: 谷氨酸; F6P: 果糖-6-磷酸; Gln: 谷氨酰胺; α-KG: α-酮戊二酸; Glucose: 葡萄糖; GAP: 3-磷酸甘油醛; PEP: 磷酸烯醇式丙酮酸; DHAP: 磷酸二羟丙酮; 3PG: 3-磷酸甘油酸。
Figure 1. Hierarchical cluster analysis-heat map of sugar catabolites in leaves of naked oat under different treatments of saline-alkali stress and NaHS spraying
CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress. Asn: asparagine; 6PG: 6-phosphogluconolactone; S7P: sedoheptulose-7-phosphate; Asp: asparate; G6P: glucose-6-phosphate; R5P: ribose-5-phosphate; FBP: fructose-1,6-diphosphate; E4P: erythrose-4-phosphate; Glu: glutamate; F6P: fructose-6-phosphate; Gln: glutamine; α-KG: α-ketoglutaric acid; GAP: 3-phosphate glyceraldehyde; PEP: enolphosphopyruvate; DHAP: phosphate dihydroxyacetone; 3PG: 3-phosphoglyceric acid.
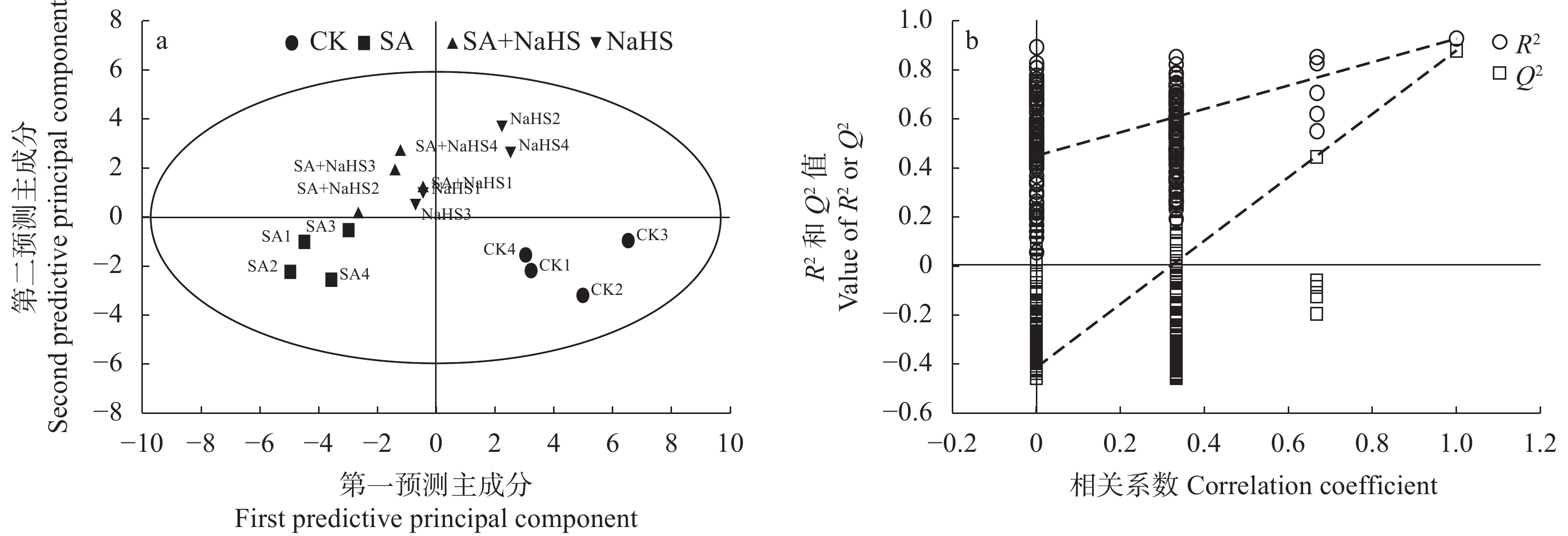
图 2 不同外源硫化氢喷施和盐碱胁迫处理下裸燕麦叶片糖分解代谢物PLS-DA得分图(a)及200次模型的置换检验(b)
CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress.
Figure 2. PLS-DA score plot (a) and 200 permutation test of the model (b) of sugar catabolites in naked oat leaves under different treatments of saline-alkali stress and NaHS spraying
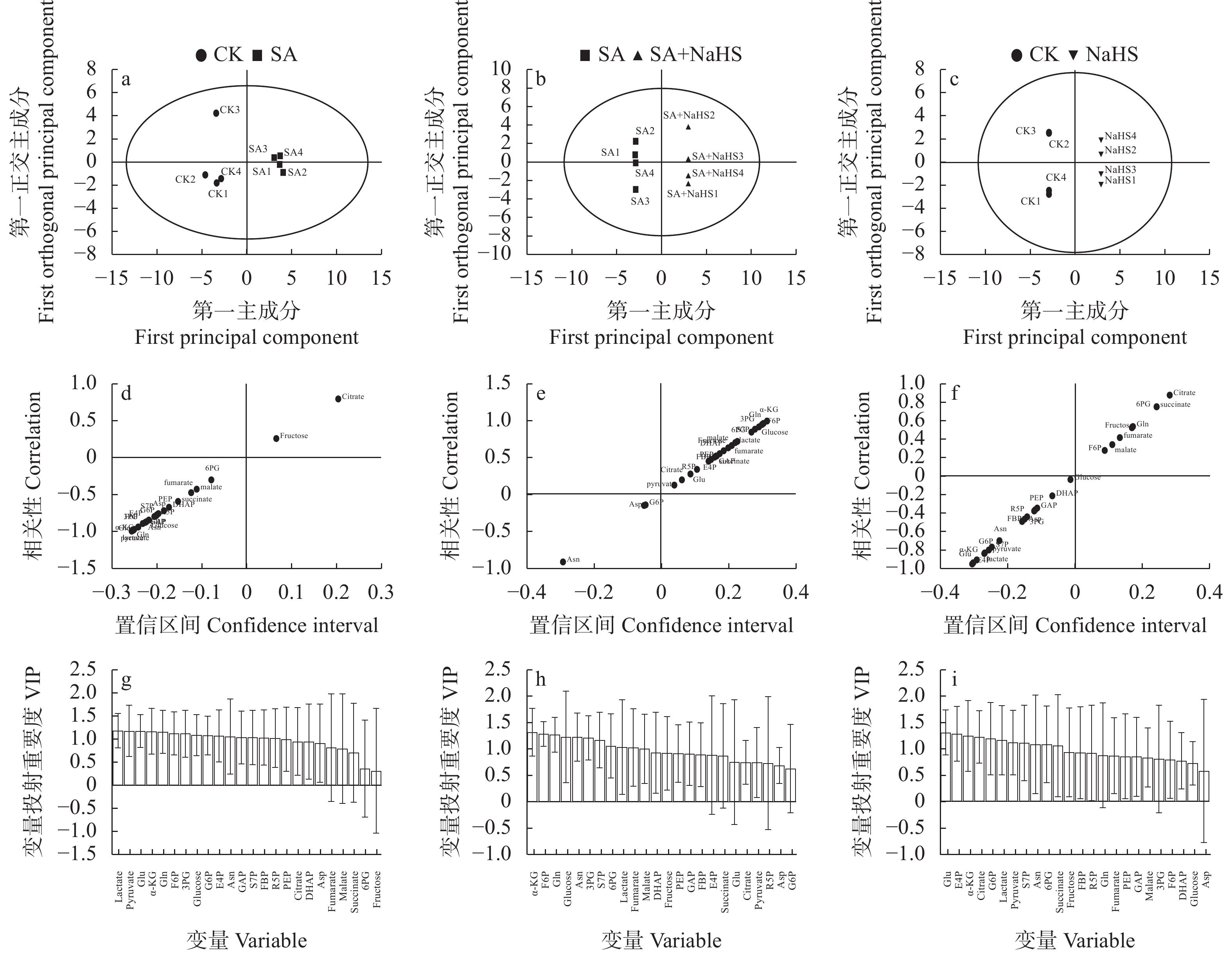
图 3 不同外源硫化氢喷施和盐碱胁迫处理下裸燕麦叶片糖分解代谢物OPLS-DA得分图(a: CK vs. SA; b: SA vs. SA+NaHS; c: CK vs. NaHS)、S形图(d: CK vs. SA; e: SA vs. SA+NaHS; f: CK vs. NaHS)和VIP图(g: CK vs. SA; h: SA vs. SA+NaHS; i: CK vs. NaHS)
CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。VIP: 变量投射重要度; 各变量名称解释说明见图1。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress. VIP: variable importance for the projection. The explainaiton of each variable is shown in the Figure 1.
Figure 3. OPLS-DA score plots (a: CK vs. SA; b: SA vs. SA+NaHS; c: CK vs. NaHS), S-plots (d: CK vs. SA; e: SA vs. SA+NaHS; f: CK vs. NaHS) and VIP plots (g: CK vs. SA; h: SA vs. SA+NaHS; i: CK vs. NaHS) of sugar catabolites in naked oat leaves under different treatments of saline-alkali stress and NaHS spraying
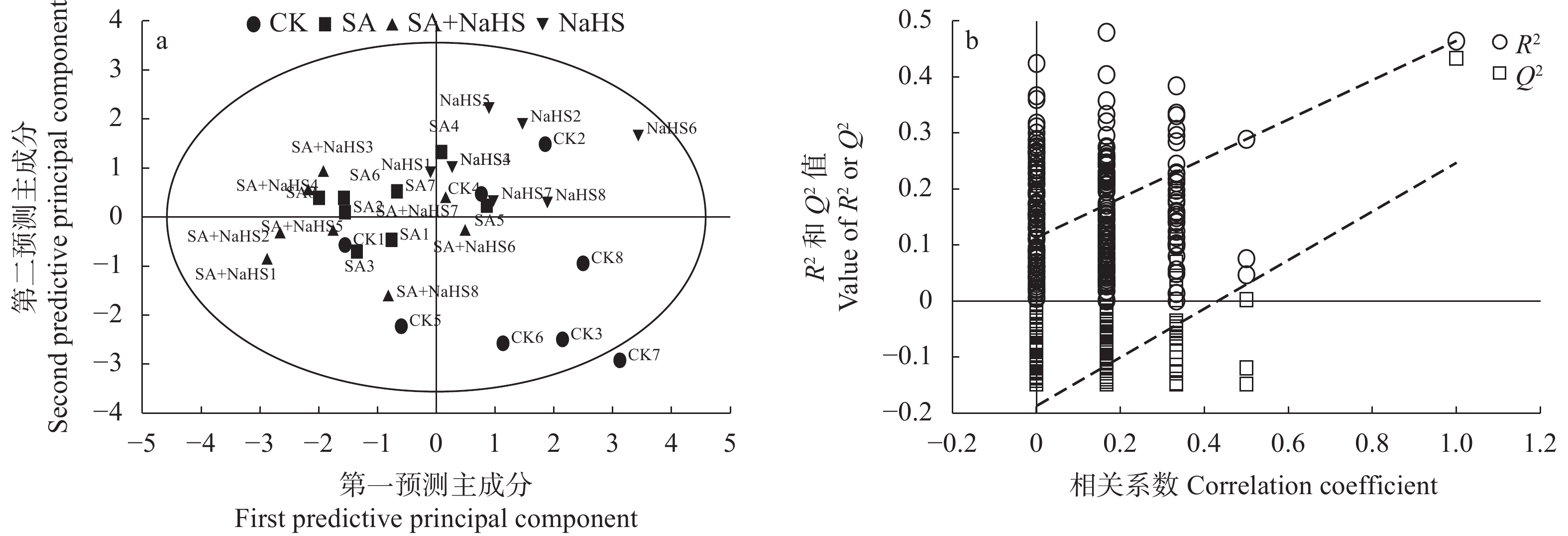
图 4 不同外源硫化氢喷施和盐碱胁迫处理下裸燕麦叶片酚酸PLS-DA得分图(a)及200次模型的置换检验(b)
CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress.
Figure 4. PLS-DA score plot (a) and 200 permutation test of the model (b) of phenolic acids in naked oat leaves under different treatments of saline-alkali stress and NaHS spraying
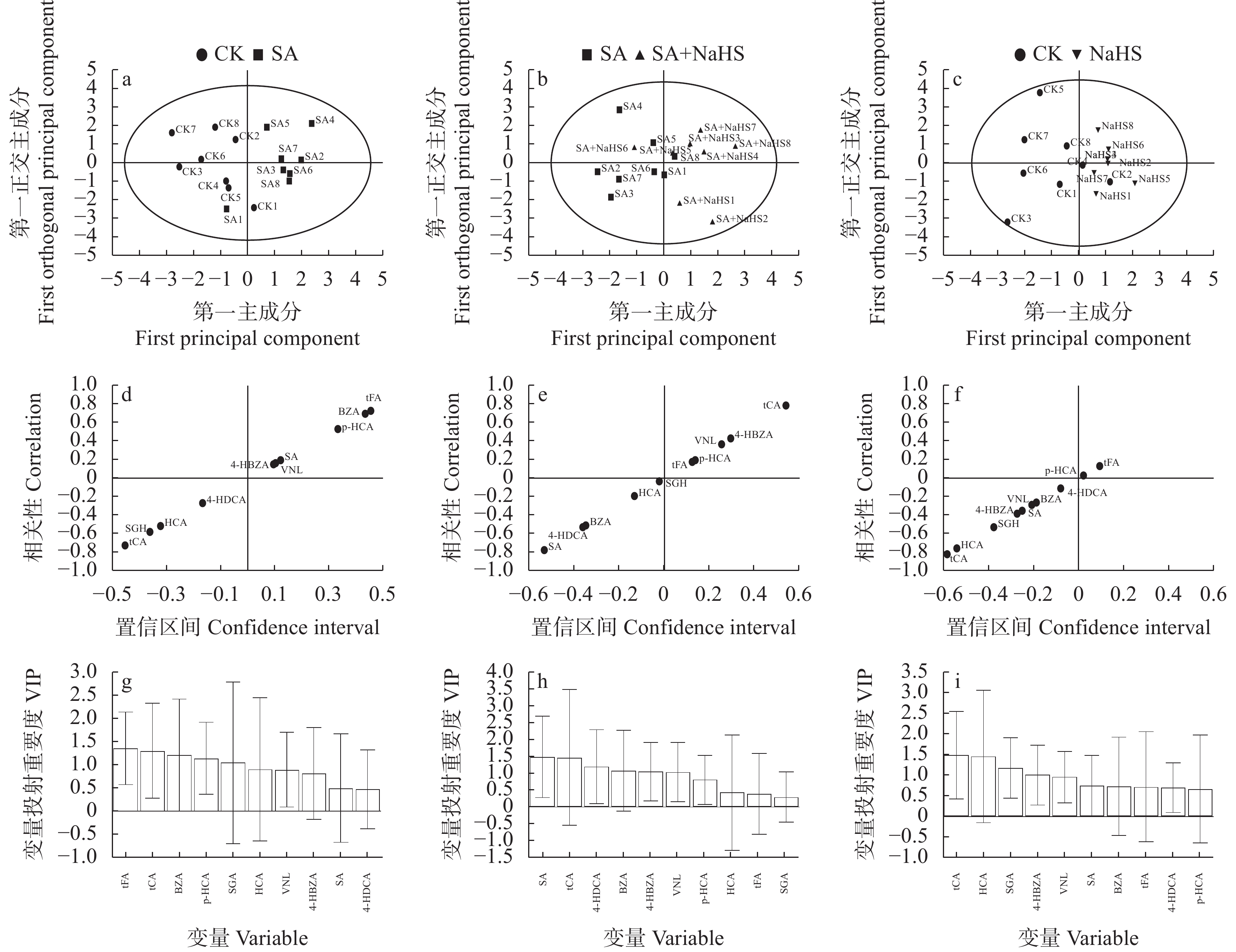
图 5 不同外源硫化氢喷施和盐碱胁迫处理下裸燕麦叶片酚酸OPLS-DA得分图(a: CK vs. SA; b: SA vs. SA+NaHSl; c: CK vs. NaHS), S形图(d: CK vs. SA; e: SA vs. SA+NaHS; f: CK vs. NaHS)和VIP图(g: CK vs. SA; h: SA vs. SA+NaHS; i: CK vs. NaHS)
CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。VIP: 变量投射重要度; GA: 没食子酸; Phe: 苯丙氨酸; DBA:原儿茶酸; PCH: 原儿茶醛; CTC: 儿茶素; VA: 香草酸; CA: 咖啡酸; SGA:丁香酸; Epi: 表儿茶素; p-HCA: 对香豆酸; 4-HBZA: 4-羟基苯甲酸; VNL: 香草醛; SGH: 丁香醛; 4-HDCA: 芥子酸; tFA: 反式阿魏酸; SA: 水杨酸; BZA: 苯甲酸; HCA: 氢化肉桂酸; tCA: 反式肉桂酸。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress. VIP: variable importance for the projection; GA: Gallic acid; Phe: Phenylalanine; DBA: 3,4-Dihydroxybenzoic acid; PCH: Protocatechualdehyde; CTC: Catechin; VA: Vanillic acid; CA: Caffeic acid; SGA: Syringic acid; Epi: Epicatechin; p-HCA: p-Hydroxycinnamic acid; 4-HBZA: 4-Hydroxybenzoic acid; VNL: Vanillin; SGH: Syringaldehyde; 4-HDCA: 4-Hydroxy-3,5-dimethoxycinnamic acid; tFA: Trans-Ferulic acid; SA: Salicylic acid; BZA: Benzoic acid; HCA: Hydrocinnamic acid; tCA: Trans-Cinnamic acid.
Figure 5. PLS-DA score plots (a: CK vs. SA; b: SA vs. SA+NaHS; c: CK vs. NaHS), S-plots (d: CK vs. SA; e: SA vs. SA+NaHS; f: CK vs. NaHS) and VIP plots (g: CK vs. SA; h: SA vs. SA+NaHS; i: CK vs. NaHS) of phenolic acids in naked oat leaves under different treatments of saline-alkali stress and NaHS spraying
图 6 OPLS-DA分析得出的外源硫化氢调控盐碱胁迫下裸燕麦叶片糖和酚酸代谢途径网络变化图
CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress.
Figure 6. Proposed metabolic pathway network changes of sugars and phenolic acids in naked oat leaves under saline-alkali stress regulated by exogenous H2S obtained from OPLS-DA analysis
表 1 外源硫化氢对盐碱胁迫下裸燕麦叶片糖代谢物含量的影响
Table 1 Effects of exogenous H2S on the contents of sugar metabolites in leaves of naked oat under saline-alkali stress
µmol∙g−1 代谢路径
Metabolic pathway代谢物
Metabolite处理 Treatment CK SA SA+NaHS NaHS 糖酵解
Glycolysis葡萄糖 Glucose 26 014.73±5023.43a 16 239.66±1357.08b 22 714.71±1929.09a 25 710.15±2980.95a 果糖 Fructose 54 486.51±8760.76a 57 984.30±8750.24a 68 690.81±8393.35a 69 090.74±17 291.12a 葡萄糖-6-磷酸 Glucose-6-phosphate 298.49±61.90a 145.73±15.64b 140.35±24.96b 162.36±38.80b 果糖-6-磷酸 Fructose-6-phosphate 65.57±8.75a 22.55±3.00c 36.69±1.84b 70.18±9.96a 果糖-1,6-二磷酸 Fructose-1,6-diphosphate 37.83±7.59a 25.02±4.23b 28.37±2.94b 30.82±6.73ab 3-磷酸甘油醛 3-Phosphate glyceraldehyde 22.27±3.39a 16.50±2.33b 19.17±2.80ab 19.96±3.75ab 磷酸二羟丙酮 Phosphate dihydroxyacetone 33.01±5.64a 25.81±3.32a 28.54±0.81a 30.38±8.13a 3-磷酸甘油酸 3-Phosphoglyceric acid 49.56±5.38a 34.95±3.03c 41.99±1.01b 45.83±3.06ab 磷酸烯醇式丙酮酸 Enolphosphopyruvate 32.86±6.05a 24.55±3.77b 28.26±3.62ab 28.98±5.20ab 丙酮酸 Pyruvate 3186.10±291.05a 1467.18±255.22c 1518.68±221.93c 2417.37±375.22b 乳酸 Lactate 71.27±7.54a 27.33±1.68d 37.62±8.58c 48.65±9.30b 戊糖磷酸途径
Pentose phosphate pathway (PPP)6-磷酸葡萄糖酯 6-Phosphogluconolactone 45.67±7.56bc 41.20±6.81c 52.62±6.27ab 58.05±4.69a 核糖-5-磷酸 Ribose-5-phosphate 60.80±14.18a 39.61±2.77b 42.90±7.03ab 49.72±9.33ab 赤藓糖-4-磷酸 Erythrose-4-phosphate 304.45±19.96a 181.31±42.78b 210.11±20.75b 220.39±14.22b 景天庚酮糖-7-磷酸
Sedoheptulose-7-phosphate46.39±5.87a 34.93±4.28b 47.89±5.29a 37.21±2.07b 三羧酸循环及相关氨基酸
Tricarboxylic acid cycle and related amino acid (TCA)柠檬酸 Citrate 1650.71±245.77b 2218.46±287.21a 2317.69±288.41a 2467.26±282.27a α-酮戊二酸 α-Ketoglutaric acid 63.48±2.78a 24.25±2.95d 54.14±2.01b 48.74±4.82c 琥珀酸 Succinate 875.27±30.09a 823.95±56.74a 939.18±144.02a 1013.98±94.26a 延胡索酸 Fumarate 445.85±104.85a 368.98±36.48a 505.78±121.97a 524.09±94.66a 苹果酸 Malic acid 3544.86±924.13a 2933.51±351.98a 3924.18±942.21a 4039.05±620.36a 谷氨酸 Glutamate 276.07±10.66a 130.63±11.95b 144.73±38.71b 174.53±24.16b 谷氨酰胺 Glutamine 1400.46±244.50ab 592.36±129.80c 1086.11±64.60b 1850.98±531.38a 天冬氨酸 Asparate 3802.91±319.93a 3419.40±319.93a 4124.16±319.93a 3874.08±319.93a 天冬酰胺 Asparagine 31.63±1.21a 35.69±1.04a 35.25±1.43a 39.46±2.60a CK: 非盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 非盐碱胁迫下喷NaHS。表中数据为平均值±标准差, 同行不同字母表示处理间在P<0.05水平差异显著。CK: no saline-alkali stress and no NaHS; SA: saline-alkali stress and no NaHS; SA+NaHS: NaHS spraying under saline-alkali stress; NaHS: NaHS spraying under no saline-alkali stress. Data are means±S.D. Different letters in the same row indicate significant differences among treatments at P<0.05 level. 表 2 外源硫化氢对盐碱胁迫下裸燕麦叶片还原型谷胱甘肽(GSH)、氧化型谷胱甘肽(GSSG)、还原型辅酶Ⅱ(NADPH)、氧化型辅酶Ⅱ(NADP)含量及比值和腺苷三磷酸(ATP)含量的影响
Table 2 Effects of exogenous H2S on the contents and ratios of reduced glutathione (GSH), oxidized glutathione (GSSG), reduced coenzyme Ⅱ (NADPH), oxidized coenzyme Ⅱ (NADP) and adenosine triphosphate (ATP) content in naked oat leaves under saline-alkali stress
处理 Treatment GSH (µmol·g−1) GSSG (µmol·g−1) GSH/GSSG NADPH (µmol·g−1) NADP (µmol·g−1) NADPH/NADP ATP (µmol·g−1) CK 27.56±5.28a 103.80±15.36a 0.268±0.048b 26.27±4.64a 14.03±2.75a 1.878±0.039a 12.22±3.05a SA 19.95±3.01b 53.81±9.86b 0.378±0.073a 19.47±2.81b 9.94±0.46b 1.961±0.281a 13.76±1.13a SA+NaHS 22.92±3.54ab 58.97±5.55b 0.387±0.028a 21.96±2.51b 11.37±1.43ab 1.934±0.037a 14.89±1.34a NaHS 23.50±3.86ab 68.27±6.27b 0.345±0.059ab 22.05±3.24b 12.08±1.97ab 1.843±0.258a 14.80±2.47a CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。表中数据为平均值±标准差, 同列不同字母表示处理间在P<0.05水平差异显著。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress. Data are means±S.D. Different letters in the same column indicate significant differences among treatments at P<0.05 level. 表 3 外源硫化氢对盐碱胁迫下裸燕麦叶片酚酸含量的影响
Table 3 Effects of exogenous H2S on the contents of phenolic acids in leaves of naked oat under saline-alkali stress
µg·g−1 酚酸 Phenolic acid 处理 Treatment CK SA SA+NaHS NaHS 没食子酸 Gallic acid 0.031±0.009b 0.043±0.015a 0.043±0.005a 0.033±0.006ab 苯丙氨酸 Phenylalanine 0.013±0.003a 0.013±0.003a 0.010±0.001a 0.013±0.006a 原儿茶酸 3,4-Dihydroxybenzoic acid 0.238±0.053b 0.278±0.060b 0.354±0.083a 0.280±0.041b 原儿茶醛 Protocatechualdehyde 1.767±0.192c 2.018±0.131b 2.282±0.427a 1.654±0.141c 儿茶素 Catechin 0.001±0.000a 0.001±0.000a 0.001±0.000a 0.001±0.000a 香草酸 Vanillic acid 10.327±0.890a 10.714±0.633a 10.883±0.901a 10.168±0.582a 咖啡酸 Caffeic acid 1.860±0.629a 1.795±0.523a 1.784±0.566a 1.882±0.502a 丁香酸 Syringic acid 4.475±0.286b 4.801±0.266a 4.643±0.235ab 4.453±0.325b 表儿茶素 Epicatechin 0.001±0.001a 0.001±0.000a 0.001±0.001a 0.001±0.001a 对香豆酸 p-Hydroxycinnamic acid 80.049±8.481b 85.333±3.229ab 86.691±7.315a 80.550±4.960b 4-羟基苯甲酸 4-Hydroxybenzoic acid 17.332±1.125ab 17.719±1.052a 18.230±0.727a 16.709±0.866b 香草醛 Vanillin 19.411±1.214b 19.943±1.382ab 20.686±1.004a 18.757±1.020b 丁香醛 Syringaldehyde 3.557±0.743a 2.947±0.510ab 2.700±1.176b 3.072±0.516ab 芥子酸 4-Hydroxy-3,5-dimethoxycinnamic acid 12.034±1.507a 11.444±1.071a 10.066±1.977a 11.759±2.360a 反式阿魏酸 Trans-Ferulic acid 162.599±11.015b 173.123±5.836a 177.174±14.843a 163.635±9.689b 水杨酸 Salicylic acid 0.579±0.115a 0.615±0.090a 0.520±0.075a 0.534±0.099a 苯甲酸 Benzoic acid 12.399±1.117bc 14.236±1.479a 13.401±1.321ab 11.900±1.361c 氢化肉桂酸 Hydrocinnamic acid 0.008±0.005a 0.005±0.002ab 0.005±0.002ab 0.004±0.001b 反式肉桂酸 Trans-Cinnamic acid 0.201±0.065a 0.145±0.018b 0.168±0.022ab 0.135±0.021b 酚酸总量 Total phenolic acid content 326.882±20.816b 345.170±9.309a 349.951±20.613a 325.538±13.128b CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷施NaHS; NaHS: 无盐碱胁迫下喷施NaHS。表中数据为平均值±标准差, 同行不同字母表示处理间在P<0.05水平差异显著。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress. Data are means±S.D. Different letters in the same row indicate significant differences among treatments at P<0.05 level. 表 4 外源硫化氢对盐碱胁迫下裸燕麦产量性状的影响(平均值±标准差)
Table 4 Effects of exogenous H2S on the yield traits of naked oat under saline-alkaline stress
处理
Treatment穗数量
Number of spike
per plant穗铃数量
Number of bolls
per spike穗粒数量
Number of grains
per spike千粒重量
1000-grain weigh
(g)单株籽粒产量
Grain yield per plant
(g)单株生物学产量
Biological yield per plant
(g)CK 6.33±0.49a 14.11±4.65a 17.73±4.37a 15.62±0.76a 1.79±0.40a 23.41±3.96a SA 5.86±1.41a 13.01±0.88a 11.05±3.03b 15.22±2.16a 1.01±0.19b 21.08±2.60a SA+NaHS 5.98±1.08a 13.96±1.74a 17.07±3.84a 14.32±0.52a 1.54±0.38a 23.10±6.25a NaHS 6.44±1.70a 16.84±2.48a 22.58±3.63a 13.76±1.15a 1.91±0.18a 24.14±2.86a CK: 无盐碱胁迫下喷水; SA: 盐碱胁迫下喷水; SA+NaHS: 盐碱胁迫下喷NaHS; NaHS: 无盐碱胁迫下喷NaHS。表中数据为平均值±标准差, 同列不同字母表示处理间在P<0.05水平差异显著。CK: no salt-alkali stress and no NaHS; SA: salt-alkali stress and no NaHS; SA+NaHS: NaHS spraying under salt-alkali stress; NaHS: NaHS spraying under no salt-alkali stress. Data are means±S.D. Different letters in the same column indicate significant differences among treatments at P<0.05 level. -
[1] LI Y Y, ZHAO K, REN J H, et al. Analysis of the Dielectric constant of saline-alkali soils and the effect on radar backscattering coefficient: a case study of soda alkaline saline soils in Western Jilin Province using RADARSAT-2 data[J]. Scientific World Journal, 2014. DOI: 10.1155/2014/563015
[2] 张毅, 石玉, 胡晓辉, 等. 外源Spd对盐碱胁迫下番茄幼苗氮代谢及主要矿质元素含量的影响[J]. 应用生态学报, 2013, 24(5): 1401−1408 ZHANG Y, SHI Y, HU X H, et al. Effects of exogenous spermidine on the nitrogen metabolism and main mineral elements contents of tomato seedlings under saline-alkali stress[J]. Chinese Journal of Applied Ecology, 2013, 24(5): 1401−1408
[3] 付寅生, 崔继哲, 陈广东, 等. 盐碱胁迫下碱地肤Na+/H+逆向转运蛋白基因KsNHX1表达分析[J]. 应用生态学报, 2012, 23(6): 1629−1634 FU Y S, CUI J Z, CHEN G D, et al. Expression of Na+/H+ antiporter gene KsNHX1 in Kochia sieversiana under saline-alkali stress[J]. Chinese Journal of Applied Ecology, 2012, 23(6): 1629−1634
[4] 闫永庆, 王文杰, 朱虹, 等. 混合盐碱胁迫对青山杨渗透调节物质及活性氧代谢的影响[J]. 应用生态学报, 2009, 20(9): 2085−2091 YAN Y Q, WANG W J, ZHU H, et al. Effects of salt-alkali stress on osmoregulation substance and active oxygen metabolism of Qingshan poplar (Populus pseudo cathayana × P. deltoides)[J]. Chinese Journal of Applied Ecology, 2009, 20(9): 2085−2091
[5] 刘建新, 刘瑞瑞, 贾海燕, 等. 外源H2S对盐碱胁迫下裸燕麦幼苗叶片渗透胁迫的调节作用[J]. 生态学杂志, 2020, 39(12): 3989−3997 LIU J X, LIU R R, JIA H Y, et al. Regulation of exogenous hydrogen sulfide on osmotic stress in leaves of naked oat seedlings under saline-alkali mixed stress[J]. Chinese Journal of Ecology, 2020, 39(12): 3989−3997
[6] LI H W, ZANG B S, DENG X W, et al. Overexpression of the trehalose-6-phosphate synthase gene OsTPS1 enhances abiotic stress tolerance in rice[J]. Planta, 2011, 234: 1007−1018 doi: 10.1007/s00425-011-1458-0
[7] 郭瑞, 周际, 杨帆, 等. 小麦根系在碱胁迫下的生理代谢反应[J]. 植物生态学报, 2017, 41(6): 683−692 doi: 10.17521/cjpe.2016.0136 GUO R, ZHOU J, YANG F, et al. Metabolic responses of wheat roots to alkaline stress[J]. Chinese Journal of Plant Ecology, 2017, 41(6): 683−692 doi: 10.17521/cjpe.2016.0136
[8] 郭家鑫, 鲁晓宇, 陶一凡, 等. 棉花在盐碱胁迫下代谢产物及通路的分析[J]. 作物学报, 2022, 48(8): 2100−2114 GUO J X, LU X Y, TAO Y F, et al. Analysis of metabolites and pathways in cotton under salt and alkali stresses[J]. Acta Agronomica Sinica, 2022, 48(8): 2100−2114
[9] 赵琦, 包玉英. 混合盐碱胁迫下丛枝菌根真菌对紫花苜蓿生长及2种酚酸含量的影响[J]. 西北植物学报, 2015, 35(9): 1829−1836 doi: 10.7606/j.issn.1000-4025.2015.09.1829 ZHAO Q, BAO Y Y. Effect of arbuscular mycorrhizal fungion growth and two phenolic acids of Medicago sativa under various mixed salt-alkaline stresses[J]. Acta Botanica Boreali-Occidentalia Sinica, 2015, 35(9): 1829−1836 doi: 10.7606/j.issn.1000-4025.2015.09.1829
[10] KAUR H, BHARDWAJ R D, GREWAL S K. Mitigation of salinity-induced oxidative damage in wheat (Triticum aestivum L.) seedlings by exogenous application of phenolic acids[J]. Acta Physiologiae Plantarum, 2017, 39(10): 221 doi: 10.1007/s11738-017-2521-7
[11] MA S Q, LV L, MENG C, et al. Integrative analysis of the metabolome and transcriptome of Sorghum bicolor reveals dynamic changes in flavonoids accumulation under saline-alkali stress[J]. Journal of Agricultural and Food Chemistry, 2020, 68: 14781−14789 doi: 10.1021/acs.jafc.0c06249
[12] GENG G, LV C H, STEVANATO P, et al. Transcriptome analysis of salt-sensitive and tolerant genotypes reveals salt-tolerance metabolic pathways in sugar beet[J]. International Journal of Molecular Sciences, 2019, 20: 5910−5928 doi: 10.3390/ijms20235910
[13] GUO R, SHI L X, YAN C R, et al. Ionomic and metabolic responses to neutral salt or alkaline salt stresses in maize (Zea mays L.) seedlings[J]. BMC Plant Biology, 2017, 17: 41−53 doi: 10.1186/s12870-017-0994-6
[14] GUO R, YANG Z Z, LI F, et al. Comparative metabolic responses and adaptive strategies of wheat (Triticum aestivum) to salt and alkali stress[J]. BMC Plant Biology, 2015, 15: 170−182 doi: 10.1186/s12870-015-0546-x
[15] 刘建新, 刘瑞瑞, 贾海燕, 等. 硫化氢对盐碱胁迫下裸燕麦叶片抗坏血酸-谷胱甘肽循环的调控效应[J]. 应用生态学报, 2021, 32(11): 3988−3996 LIU J X, LIU R R, JIA H Y, et al. Regulation effects of hydrogen sulfide on ascorbate-glutathione cycle in naked oat leaves under saline-alkali stress[J]. Chinese Journal of Applied Ecology, 2021, 32(11): 3988−3996
[16] LAI D W, MAO Y, ZHOU H, et al. Endogenous hydrogen sulfide enhances salt tolerance by coupling the reestablishment of redox homeostasis and preventing salt-induced K+ loss in seedlings of Medicago sativa[J]. Plant Science, 2014, 225: 117−129 doi: 10.1016/j.plantsci.2014.06.006
[17] SUN Y P, MA C, KANG X, et al. Hydrogen sulfide and nitric oxide are involved in melatonin-induced salt tolerance in cucumber[J]. Plant Physiology and Biochemistry, 2021, 167: 101−112 doi: 10.1016/j.plaphy.2021.07.023
[18] CHEN J, WANG W H, WU F H, et al. Hydrogen sulfide enhances salt tolerance through nitric oxide-mediated maintenance of ion homeostasis in barley seedling roots[J]. Scientific Reports, 2015, 5: 12516 doi: 10.1038/srep12516
[19] MOSTOFA M G, SAEGUSA D, FUJITA M, et al. Hydrogen sulfide regulates salt tolerance in rice by maintaining Na+/K+ balance, mineral homeostasis and oxidative metabolism under excessive salt stress[J]. Frontiers in Plant Science, 2015, 6: 1055 doi: 10.3389/fpls.2015.01055
[20] 黄菡, 郭莎莎, 陈良超, 等. 外源硫化氢对盐胁迫下茶树抗氧化特性的影响[J]. 植物生理学报, 2017, 53(3): 497−504 HUANG H, GUO S S, CHEN L C, et al. Effects of exogenous hydrogen sulfide on the antioxidant characteristics of tea plant (Camellia sinensis) under salt stress[J]. Plant Physiology Journal, 2017, 53(3): 497−504
[21] SHAN C, LIU H, ZHAO L, et al. Effects of exogenous hydrogen sulfide on the redox states of ascorbate and glutathione in maize leaves under salt stress[J]. Biologia Plantarum, 2014, 58(1): 169−173 doi: 10.1007/s10535-013-0366-5
[22] 郑州元, 林海荣, 崔辉梅. 外源硫化氢对盐胁迫下加工番茄幼苗光合参数及叶绿素荧光特性的影响[J]. 核农学报, 2017, 31(7): 1426−1435 ZHENG Z Y, LIN H R, CUI H M. Effect of exogenous hydrogen sulfide on photosynthesis parameters and chlorophyll fluorescence characteristics of processing tomato (Lycopersicon esculentum Mill ssp. subspontaneum Brezh) seedlings under NaCl stress[J]. Journal of Nuclear Agricultural Sciences, 2017, 31(7): 1426−1435
[23] MONTESINOS-PEREIRA D, DE LA TORRE-GONZÁLEZ A, BLASCO B, et al. Hydrogen sulphide increase the tolerance to alkalinity stress in cabbage plants (Brassica oleracea L. ʻBroncoʼ)[J]. Scientia Horticulturae, 2018, 235: 349−356 doi: 10.1016/j.scienta.2018.03.021
[24] 郑殿升, 张宗文. 大粒裸燕麦(莜麦) (Avena nuda L.)起源及分类问题的探讨[J]. 植物遗传资源学报, 2011, 12(5): 667−670 ZHENG D S, ZHANG Z W. Discussion on the origin and taxonomy of naked oat (Avena nuda L.)[J]. Journal of Plant Genetic Resources, 2011, 12(5): 667−670
[25] GAO W Y, FENG Z, BAI Q Q, et al. Melatonin-mediated regulation of growth and antioxidant capacity in salt-tolerant naked oat under salt stress[J]. International Journal of Molecular Sciences, 2019, 20(5): 1176 doi: 10.3390/ijms20051176
[26] 刘建新, 刘瑞瑞, 刘秀丽, 等. 不同时期喷施NaHS对盐碱胁迫下裸燕麦叶片渗透调节物质和抗氧化活性的影响[J]. 生态学杂志, 2021, 40(11): 3620−3632 LIU J X, LIU R R, LIU X L, et al. Effects of spraying NaHS at different growth stages on osmotic adjustment substance and antioxidant activity in leaves of naked oat under saline-alkali stress[J]. Chinese Journal of Ecology, 2021, 40(11): 3620−3632
[27] 高龙飞, 贾斌, 张卫华, 等. 盐胁迫下蓝莓叶片生理特性与代谢组学分析[J]. 植物生理学报, 2022, 58(1): 155−164 doi: 10.13592/j.cnki.ppj.2021.0287 GAO L F, JIA B, ZHANG W H, et al. Physiological characteristics and metabonomics analysis of blueberry leaves under salt stress[J]. Plant Physiology Journal, 2022, 58(1): 155−164 doi: 10.13592/j.cnki.ppj.2021.0287
[28] 陈晓晶, 徐忠山, 赵宝平, 等. 盐胁迫对燕麦根系呼吸代谢、抗氧化酶活性及产量的影响[J]. 生态学杂志, 2021, 40(9): 2773−2782 doi: 10.13292/j.1000-4890.202109.036 CHEN X J, XÜ Z S, ZHAO B P, et al. Effects of salt stress on root respiratory metabolism, antioxidant enzyme activities, and yield of oats[J]. Chinese Journal of Ecology, 2021, 40(9): 2773−2782 doi: 10.13292/j.1000-4890.202109.036
[29] LIU H, WANG J C, LIU J H, et al. Hydrogen sulfide (H2S) signaling in plant development and stress responses[J]. Abiotech, 2021, 2: 32−63 doi: 10.1007/s42994-021-00035-4
[30] WEI M Y, LIU J Y, LI H, et al. Proteomic analysis reveals the protective role of exogenous hydrogen sulfide against salt stress in rice seedlings[J]. Nitric Oxide, 2021, 111/112: 14−30 doi: 10.1016/j.niox.2021.04.002
[31] JIANG J L, REN X M, LI L, et al. H2S Regulation of metabolism in cucumber in response to salt-stress through transcriptome and proteome analysis[J]. Frontiers in Plant Science, 2020, 11: 1283 doi: 10.3389/fpls.2020.01283
[32] KOVÁCS Z, SIMON-SARKADI L, SOVÁNY C, et al. Differential effects of cold acclimation and abscisic acid on free amino acid composition in wheat[J]. Plant Science, 2011, 180(1): 61−68 doi: 10.1016/j.plantsci.2010.08.010
[33] ZHAO Y, XU J Y, HE L, et al. Sugar-induced tolerance to the salt stress in maize seedlings by balancing redox homeostasis[J]. Agriculture Forestry and Fisheries, 2016, 5(4): 126−134 doi: 10.11648/j.aff.20160504.15
[34] 孙晓莉, 田寿乐, 沈广宁, 等. 干旱胁迫下H2S对板栗幼苗根系抗氧化特性及呼吸相关酶活性的影响[J]. 核农学报, 2019, 33(5): 1024−1031 SUN X L, TIAN S L, SHEN G N, et al. Effect of exogenous hydrogen sulfide on antioxidant characteristics and respiratory related enzymes in root of chestnut seedlings under drought stress[J]. Journal of Nuclear Agricultural Sciences, 2019, 33(5): 1024−1031
[35] LINIĆ I, ŠAMEC D, GRÚZ J, et al. Involvement of phenolic acids in short-term adaptation to salinity stress is species-specific among Brassicaceae[J]. Plants, 2019, 8(6): 155 doi: 10.3390/plants8060155
[36] BANIK N, BHATTACHARJEE S. Complementation of ROS scavenging secondary metabolites with enzymatic antioxidant defense system augments redox-regulation property under salinity stress in rice[J]. Physiology and Molecular Biology of Plants, 2020, 26(8): 1623−1633 doi: 10.1007/s12298-020-00844-9
-
期刊类型引用(2)
1. 刘胜红,杨清,吴化雨,唐勇,王芳,李根前,邓佳. 羧甲基壳聚糖诱导葡萄柚果实抗病防御代谢物差异分析. 西南林业大学学报(自然科学). 2024(02): 163-173 .  百度学术
百度学术
2. 王维轩,李世洁,梁继杰,王秦政,苏畅,高克明,李宗明,吕昊峰,梁斌,周伟伟. 硫化氢调控酚类化合物代谢缓解番茄盐胁迫的作用机制. 植物生理学报. 2024(03): 518-528 .  百度学术
百度学术
其他类型引用(5)



 下载:
下载: